Discussion
Home ‣ Chemical Engineering ‣ Petroleum Refinery Engineering See What Others Are Saying!
- Question
A fuel oil consists of 4 fractions A, B, C and D. Their molar compositions and vapour pressures are given below :
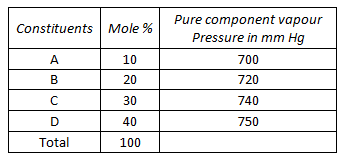
The vapour pressure of the fuel oil will be __________ mm Hg.
Options- A. 736
- B. 727.5
- C. 512
- D. 767.8
- Correct Answer
- 736
- 1. Which of the following efficiencies can be greater than 100%?
Options- A. Overall plate efficiency.
- B. Murphree plate efficiency.
- C. Point efficiency.
- D. None of these. Discuss
- 2. Which of the following nuclear reactors is the most efficient thermodynamically while operating between the same temperature and pressure limits of the reactor?
Options- A. Molten sodium cooled
- B. CO2 gas cooled
- C. Pressurised water
- D. Boiling water Discuss
- 3. Which of the following undergoes fission reaction easily?
Options- A. U-235
- B. U-238
- C. Th-232
- D. none of these Discuss
- 4. Which of the following is not used as a moderator in nuclear reactor?
Options- A. Molten sodium
- B. Light water
- C. Beryllium
- D. Boron hydride Discuss
- 5. Which of the following nuclear materials is fissile?
Options- A. Uranium-238
- B. Thorium-232
- C. Plutonium-239
- D. None of these Discuss
- 6. In the nuclear reaction, 93N239→ 94Pu239 +?; the missing particle is a/an
Options- A. electron
- B. proton
- C. neutron
- D. position Discuss
- 7. Which of the following ores contains maximum percentage of uranium?
Options- A. Carnotite
- B. Thorium.
- C. Rescolite
- D. Pitchblende Discuss
- 8. The atomic weight and atomic number of an element are A and Z respectively. What is the number of neutrons in the atom of that element?
Options- A. A+Z
- B. A- Z
- C. A
- D. Z Discuss
- 9. Which of the following isotopes is not present in natural uranium?
Options- A. U-238
- B. U-234
- C. U-235
- D. U-232 Discuss
- 10. Which of the following is present in Bhabha Atomic Research Centre (BARC) complex, Trombay (Bombay)?
Options- A. Power reactor
- B. Research reactor
- C. Fast breeder reactor
- D. Heavy water plant Discuss
More questions
Correct Answer: Murphree plate efficiency.
Correct Answer: Boiling water
Correct Answer: U-235
Correct Answer: Molten sodium
Correct Answer: Plutonium-239
Correct Answer: electron
Correct Answer: Pitchblende
Correct Answer: A- Z
Correct Answer: U-232
Correct Answer: Research reactor
Comments
There are no comments.More in Chemical Engineering:
Programming
Copyright ©CuriousTab. All rights reserved.
